Blog
An unexpected trip to Curaçao
“How would you like to go to Curaçao?” These were not the words I expected to hear when I answered my phone after the third consecutive call from an “Unknown Number” on Monday night. Thirty-six hours later, I was dragging a heavy metal object through the airport on my way to join the search for shark dermal denticles. My surprise journey with the Grab Sampler is only part of what is becoming an epic battle against Murphy’s Law in the ongoing Denticle Quest of the Baseline Caribbean team. This post details some of the obstacles the team has already overcome. You can watch many of these events unfold in the live-science videos.
Obstacle 1: Small Denticles
Shark dermal denticles are small—smaller than a grain of salt—and you might think that collecting denticles would be harder than finding a needle in a haystack. However, their small size helps the Baseline Caribbean team narrow down potential denticle sites. In areas washed by waves or current, any denticles would likely be swept away. Consequently, denticles only accumulate in low-energy locations where they can settle undisturbed. These sheltered locations are what the team is seeking in “1: Looking for Sites.” Silty sediment is an indication that the water is quiet enough for the denticles to remain.
Obstacle 2: Deep Denticles
The team came prepared to scuba dive in and dig for denticles by hand. You can see them at work in “2: The Perfect Sediment.” However, it turns out that in Curaçao at least, much of the “perfect sediment” is deeper than the divers are prepared to go. So, how to collect these deep silty sediments without diving? The Grab Sampler provides a solution: it is a heavy metal scooper that can be lowered over the side of a boat to collect deep sediments. Problem solved, right? …except that the Grab Sampler remained in Panamá. Lucky for me, the easiest way to get it to Curaçao was for me to bring it. I got to travel to Curaçao, several customs officers got to learn about the mysterious metal object in my luggage, and the Baseline Caribbean team got back to work, digging denticles.
Obstacle 3: The Grab Sampler works! And then it doesn’t.
The Grab Sampler may look like a boring metal scoop, but it is actually an instrument of high drama and suspense. After it’s lowered over the side of the boat, it sinks into the blue depths and disappears. The line keeps feeding downward, down past the rising bubbles, down farther until suddenly it goes slack: the Grab has hit bottom. Quickly, all hands go to the rope, everyone sweating to pull it back up and wondering what it will bring. That first retrieval, we all crowded around, watching as it surfaced, watching as Felix swung it over the side, leaning in to see… a single algae-encrusted shell deposited on the tray. The second go brought nothing. We tried a slower descent, than a faster one. We moved shallower, then deeper. Finally, we started bringing up the silty material we were looking for. Success! We danced the Denticle Dance, and our collection of sample bags started growing.
Unfortunately, that was not the end of the Grab Sampler’s drama. As we sent it down again, the wind picked up, pushing the boat over the descending line. The Grab Sampler must have dragged along the bottom, because when we retrieved it the sturdy metal was bent out of shape, rendering the instrument useless. As we research options for fixing the Grab, the team is back to diving for sediment in the sites that are not too deep.
. . .
Beyond these obstacles, the team has struggled against 75 million-year-old radiolarians (the backbone of the island is silica-rich rock made of ancient plankton skeletons, and as it erodes into the bays it makes the denticle search more difficult), wary property owners, Carnival traffic, difficult-to-find docks, and even an issue with the coast guard. Yet even when it seems that everything that can go wrong does go wrong, the team finds a way around the problem and the search for denticles continues. This is how science works—often it is the challenges that lead to creative problem solving, novel approaches, and interesting new questions. And sometimes when things go wrong, an intern gets to go on an unexpected trip to Curaçao.
5: Looking for fossils
4: A good day and a bad day
2: The perfect sediment
1: Looking for sites
Landed!
New expedition: Curaçao February 20th
The Baseline Caribbean team is gearing up for another expedition, this time to the sparkling, cerulean seas of the Netherlands Antilles in the Southern Caribbean.
Reefs on these Dutch Islands are in relatively good shape when compared to the rest of the Caribbean. But what were they like in the past?
Anecdotal evidence suggests that sharks were once abundant around these islands, yet empirical data are desperately needed to guide management. Reef fish communities are overfished today, but by how much? When did coral communities begin to deteriorate, and did it depend on their proximity to historical settlements?
To get at these questions and more, we plan to collect nearly a ton of sediment from modern and fossil reefs along the southwest coast of Curaçao. We also hope to get the chance to explore Klein Curaçao — a 1.7-square-kilometer uninhabited island just southeast of its big sister and namesake.
Instead of reading about the results in a stale journal in two years’ time, experience science in action. Beginning February the 20th, join us with daily posts, photos, and short videos from the field on the Baseline Caribbean science blog.
This expedition builds on our previous exploits in Panama, Belize and the Dominican Republic. We see familiar faces return: Erin Dillon (who recently hightailed it to the McCauley Lab), faithful malacologist Felix Rodriguez and ring-leader Aaron O’Dea. We are also joined by some fresh blood in the form of fish ecologist and evolutionary biologist Michele Pierotti and STRI videographer extraordinaire Ana Endara.
A huge Thank You to our supporters who will make it possible: The Caribbean Research and Management of Biodiversity field station (CARMABI) who kindly gave us a Research Prize, the Smithsonian Tropical Research Institute (STRI), and YOU! – the generous donors who contributed to our crowd-funding campaign. Stay tuned…!

When parrotfish abound the reef grows faster
Several years ago Katie Cramer, Dick Norris and I hatched a plan. We knew that the sediments on coral reefs preserved the robust teeth of fish and we guessed that the branching corals of the reef would hold it all in place. We just needed a way to extract the layers of reef sediments to reconstruct the history of fish communities on the reef.
Katie led the project with support from MarineGEO and Dick built a pushcore/vibracore hybrid, with which we managed to extract a good number of 3-4 metre long cores from coral reefs in Bocas del Toro, Panama.
Back at Scripps Katie led the troops to split the cores, take samples along them and extract the teeth from the matrix by acid digestion of the carbonate sediments. Sounds easy?
Then she had to identify what all these teeth were. Along with the team in Norris’ lab, and help from our lab in Panama, she built a reference collection of coral reef fish teeth, which turn out to be variable in shape, but on the whole extremely well-preserved over millennia.

We needed to date the cores, that’s where Jian-xin Zhao at University of Queensland came in. There we were able to date small pieces of coral using the U-Th dating technique which gave really high-resolution dates, and more importantly showed that the chronology of the cores were intact – i.e. there had been no mixing up and down which would have ruined any attempt to explore changes through time.
Our cores from Bocas stretched back 3000 years or so, and one of the most abundant teeth that Katie found was the various teeth produced by parrotfish.
Using the dates of the samples to calculate reef accretion rates we discovered that as the reef was growing it did so at a faster rate when there were more parrotfishes. This results shows that the benefit of parrotfishes for the health of the reef is always high, not only in the degraded habitats of today but also on “near-pristine” reefs which were much less fished.

The fossil record is a powerful tool to reveal ecological processes that have direct implications for conservation. And parrotfish conservation must be made a priority for the recovery and persistence of coral reefs.
Denticle side view
Some of the denticles that we find in the sediments are exceptionally well-preserved. Here is an example of a denticle that still possesses the basal plate that once held it to the underlying skin. In many cases, though, we only find the crown (the top part). This side view taken via scanning electron microscopy (SEM) can give you an idea of the three-dimensionality of a fully intact denticle. It almost looks like some strange sort of hat (which perhaps I will 3D-print, patent, and sell online if our funding runs out prematurely or is frozen by the government… just kidding).

Some things you may notice about this denticle:
- It has a very thick crown proportional to the crown length. While many fast swimming shark species have denticles that are thin and light, this denticle means business. Its thickness lends durability, forming a protective ‘armor’ around the shark. This type of denticle is generally found on sharks that live in close-association with the benthos.
- The crown is positioned at a slight angle relative to the base. This angle can alter the way in which water flows over the denticle, thereby changing the hydrodynamics of the shark on a micro level. Some of the fastest swimming species are thought to be able to vary this angle through adjustments to the basal plate and skin tension (Raschi & Tabit 1992).
Literature cited
Raschi W, Tabit C (1992) Functional aspects of placoid scales: a review and update. Aust J Mar Freshw Res 43:123–147
Evidence of a nighttime visitor

Scanning electron microscope image of a denticle extracted from the fossil reef in Bocas del Toro, Panama. 283x magnification.
Here is a denticle that I was not expecting to find. It belongs to a shark that I only on second thought included in my denticle reference collection, one that ought not belong on a coral reef. This appears to be the denticle of a crocodile shark (Pseudocarcharias kamoharai). While denticles can normally only be identified at the family level, this one fails to resemble any other denticle in my reference collection… except one. It looks almost identical to those that I isolated from the skin of a verified crocodile shark at the Smithsonian National Museum of Natural History (image below), except that it is a little smaller and much more weathered.

Scanning electron microscope image of a denticle isolated from the body of a crocodile shark for inclusion in our reference collection. 185x magnification.
Not only are crocodile sharks unassociated with coral reefs – in fact they generally only ascend from the depths at night to feed – but this shark has not been documented in the region in which this denticle was found. What’s more amazing is that this denticle is not even modern. It was extracted from our 6,600-year-old fossil reef!
While you may have never heard of a crocodile shark, they are listed as ‘Near Threatened’ on the the International Union for Conservation of Nature (IUCN) Red List. We obviously need to learn more about this species of shark in order to better protect it.
Our finding suggests that crocodile sharks did occasionally venture onto the lagoonal reefs of Bocas del Toro, Panama in the past (and perhaps even in the present day as well). More importantly, to me, this denticle epitomizes the beauty of the technique. Extracting and analyzing dermal denticle assemblages can reveal the rare, cryptic, or ephemeral elasmobranch visitors on reefs, an otherwise very challenging task to accomplish.

Crocodile shark. Source: PIRO-NOAA Observer Program. Wikimedia commons.
Time to ring in the New Year with some coral measurements
Sometimes we do a little more than just dig in the sand. Over the New Year, Mauro and I taught the coral reef ecology unit of an undergraduate field course from the University of Wisconsin–Green Bay and St. Norbert College at the STRI field station in Bocas del Toro, Panama. While we spent a portion of our time in the lab discussing the importance of the area and looking at creatures under microscopes, we also brought the class snorkeling on the reefs with us to do some field work and explore. For some of the students, this was their first time snorkeling and seeing a coral reef in person. For me, that moment was life-changing, so I’m delighted to be able to share this magic with them. We visited a diversity of reefs during the trip, some of which were clearly healthier than others, and we discussed the differences between them and the potential underlying anthropogenic and natural drivers. For example, instead of merely reading that runoff and eutrophication are harmful for coral reefs, the students were able to glimpse algae-covered corals off the coast of the Changuinola River floodplain, the site of extensive banana plantations.
The students also got some hands-on experience doing field work. With the help of the class, we’re investigating long-term bioerosion rates on branching corals, specifically staghorn (Acropora cervicornis) and finger (Porites sp.) corals, in Almirante Bay. This study will improve our understanding of the carbonate budget on reefs in the region and, in particular, which way the seesaw between reef accretion and dissolution is tilting. This question pertains directly to the future of reefs in Bocas del Toro as well as how resilient they may be to future perturbations such as storms. Furthermore, it helps us key into one of the processes contributing to the reef substrate, which is where our lab collects bulk samples and sediment cores to reconstruct historical reef communities. Understanding whether the reef is actively contributing to this substrate through coral growth and reef accretion or whether it is stagnant or even shrinking can provide context for interpreting patterns of microfossil abundance. This study may also shed light on dates we computed for corals extracted from cores at these sites, which suggest that some of the dead corals lying on the surface are far from modern.
We started this study in December 2015 by collecting, measuring, and redeploying recently-dead pieces of coral on mesh plots. We successfully relocated the plots this past December and took our first annual measurements. Some bioeroders were even see in action on the reef! Interesting trends are already starting to emerge in the data. One of the students will be conducting an independent study to try to unravel some of these patterns, so stay tuned for her findings.
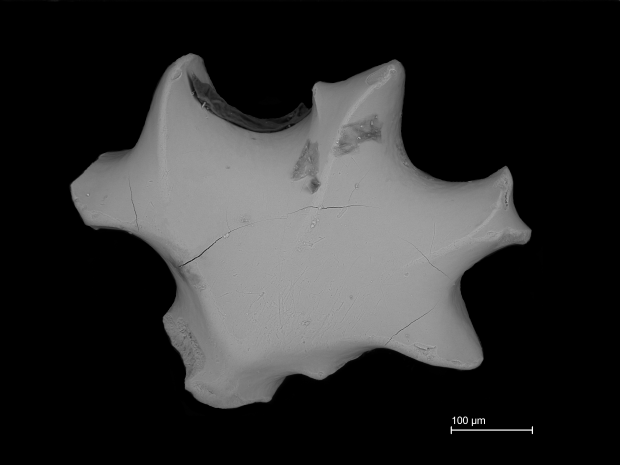
Twins in time

At first glance these two shark dermal denticles appear quite similar, right? Sure, one is a little wider than the other, but they both have distinct peaks, ridges, and intriguing honeycombed ornamentation on their crowns. What if I was to tell you that they are separated in time by about 7,000 years. Which do you think came from the fossil reef?
The next installment of shark denticle photos is here!
Over the next several weeks, I’ll be showcasing scanning electron microscope (SEM) images of some of the denticles that we extracted from the modern and ~7,000-year-old fossil coral reefs in Bocas del Toro, Panama. While the SEM images that I released previously were from known species of sharks in my reference collection, the denticles that I’m about to show you came from sediment samples we collected and processed. Our job now is to become sleuths and figure out what types of shark shed them.
Like a portrait, these denticles can paint a picture of the sharks they came from. They are the bards of the sharks of lore, lost in the sands of time… Ok, that’s one massive cliché (and I might be a bit too obsessed with denticles), but you get the point. Denticle morphology can provide us with useful insight into the historical ecology of sharks. For example, a denticle’s thickness can reveal whether its owner lived in the crevices of a reef or up in the water column. Its ridges can tell us whether or not it was a fast swimmer, speed which it may have used to catch nimble prey or swim long distances.
Some denticles are better storytellers than others, however, so we need to figure out who is who. This is one of the more unusual denticles that I’ve found so far. What type of shark do you think it may have come from? Stay tuned for my interpretation.
The third iteration of sediment processing
The sediment processing workshop will now resume in a new location: UC Santa Barbara. First things first, I had to transform the lab. Since the size of each sample was smaller, I could also downsize the set-up a bit and move everything indoors. This meant no more massive outdoor ‘drying bubbles’ or tent-covered mazes of 5-gal buckets for digesting sediments… at least for now. We did, however, order 100 gallons of acetic acid. What a lot of vinegar!
The funnest challenge was building the new drying oven from an old storage cabinet, several power strips, and a box full of incandescent light bulbs. Drilling through the metal cabinet to feed the cords through was by far the hardest part. Standard drill bits didn’t cut it, so I had to upgrade to a step drill bit. That worked like a charm, but it still took several hours of drilling. Once everything was installed, I turned on the power strips and basked in the glow for a moment. It was toasty – exactly as intended! When I did a test run the next morning, I was pleased to find that the sediment dried in under a day.
Now, we are just about ready to rock and roll…









